Project Overview
As a Medical Device Design Engineer at Alcon for three years, I managed the lifecycle of next-generation intraocular lens delivery systems. My role bridged the gap between rapid iterative design and rigorous quality engineering, ensuring devices met strict FDA standards through data-driven validation.
Technical Contributions
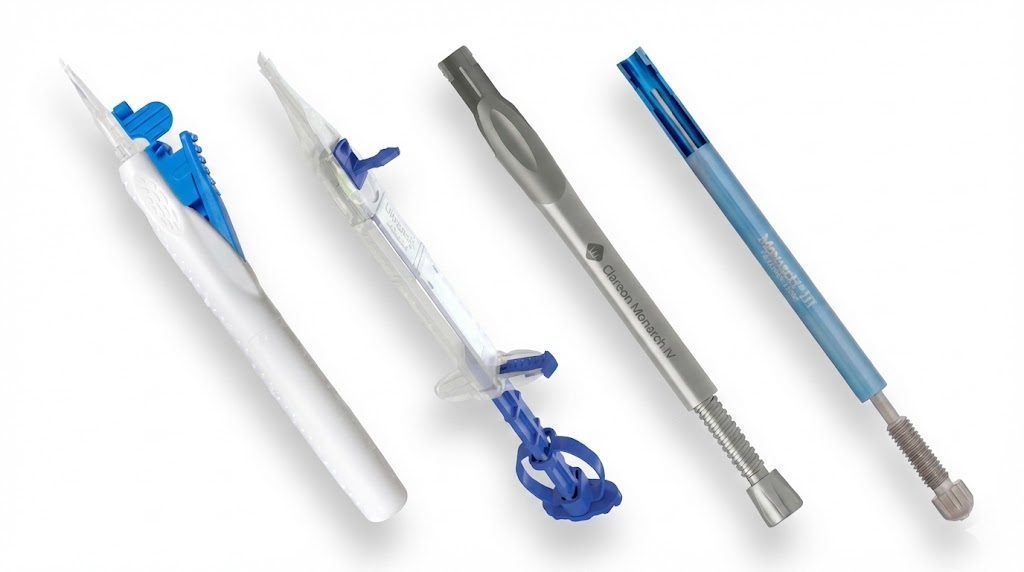
1. Rapid Prototyping & DFM
3D CAD concepts with PTC Creo for mechanical design and 3D printing for immediate validation.
- Transformed functional prototypes into mass-producible designs by performing Design for Manufacturing (DFM) analysis for injection molding.
- Collaborated directly with manufacturing vendors to optimize tool design, ensuring complex geometries could be molded consistently at scale.
2. Statistical Analysis & Validation
Moved beyond “pass/fail” testing by implementing rigorous statistical quality control using Minitab:
- Defined delivery testing requests and executed hypothesis testing (e.g., 2-sample % defective tests) to validate design changes against legacy standards.
- Utilized statistical models to analyze failure rates, ensuring the final product met Six Sigma reliability requirements before transfer to manufacturing.
3. Human-Centered Design
Organized and led clinical studies with ophthalmic surgeons to refine device ergonomics.
- Translated qualitative feedback from wet labs into quantitative engineering requirements.
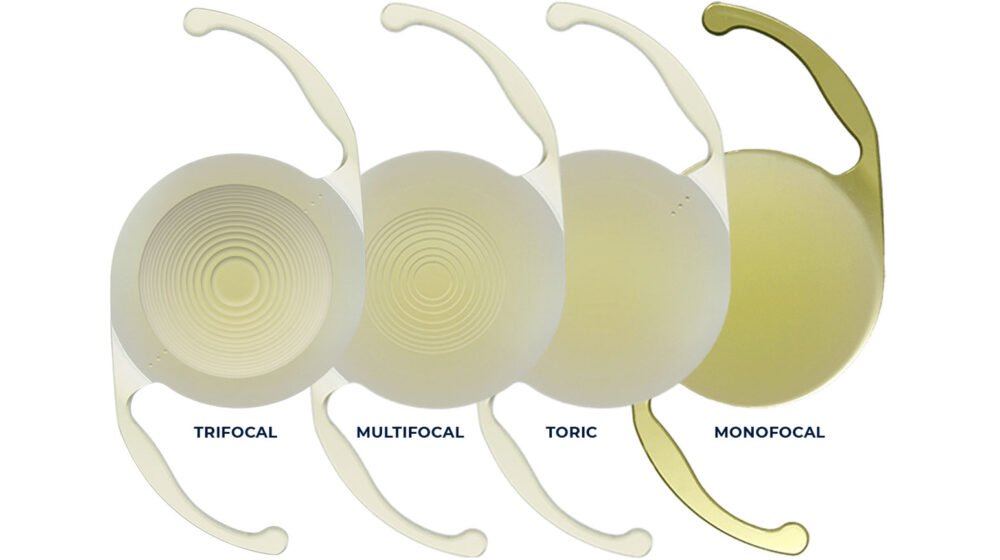
- Balanced user needs (tactile feel, ease of use) with strict mechanical tolerances required for the intraocular lens protection.